SCEP/NDES Sunset Strategy for Healthcare
The core SCEP/NDES sunset strategy covers the replacement landscape, device compatibility matrix, and parallel-run migration pattern. Healthcare adds constraints that make SCEP migration uniquely difficult: FDA-regulated devices that can't be modified, clinical mobile devices with specific MDM requirements, and legacy clinical systems with hardcoded enrollment paths.
Medical Device Certificate Enrollment: The SCEP Dependency
Connected medical devices — infusion pumps, patient monitors, imaging systems (MRI, CT, X-ray), laboratory analysers, ventilators — are the SCEP stronghold. Many of these devices run embedded operating systems with certificate enrollment capabilities limited to SCEP. There's no firmware update path to EST or ACME.
The device lifecycle is the core constraint. Medical devices in active clinical use often remain deployed for 10-15 years. A device manufactured in 2015 with SCEP-only enrollment will be in service until 2025-2030. There's no business case or clinical case for replacing a functioning $500,000 MRI scanner because its certificate enrollment protocol is outdated.
PKI Health Radar
Drag the sliders to assess your current posture — scores update instantly.
For these devices, the strategy isn't migration — it's containment. Segment SCEP-dependent medical devices onto dedicated network segments. Harden NDES with per-device challenge passwords where the device supports it. Monitor enrollment activity for anomalies. And document the risk acceptance in your HIPAA risk assessment.
FDA-Cleared Devices and the Firmware Update Constraint
The FDA's 510(k) clearance process validates a medical device's safety and effectiveness — including its software. Modifying the firmware of an FDA-cleared device to support a new enrollment protocol may constitute a design change that requires re-submission and re-clearance. Depending on the nature of the change, this can take months to years.
This means device manufacturers, not healthcare organisations, control the pace of protocol modernisation. Even when a manufacturer releases a firmware update with EST support, the healthcare organisation must validate the update, schedule deployment during maintenance windows (which in clinical environments are rare and short), and coordinate with the device's biomedical engineering team.
The practical implication: for FDA-regulated devices, SCEP sunset timelines are measured in manufacturer release cycles and clinical deployment windows, not IT project timelines. Expect 3-5 years from manufacturer availability to complete fleet migration for a single device type.
Clinical Mobile Device Provisioning
Clinicians increasingly use mobile devices — tablets for bedside charting, smartphones for clinical communication, dedicated mobile devices for point-of-care testing. These devices need certificates for Wi-Fi (EAP-TLS), VPN, and application access.
Most clinical MDM deployments use SCEP via a connector to ADCS/NDES for certificate provisioning. Modern MDM platforms (Intune, Jamf, VMware Workspace ONE) support EST and ACME, making these devices the first candidates for SCEP migration.
The challenge is clinical workflow continuity. Certificate re-provisioning must happen without clinician intervention — a nurse discovering their tablet can't connect to Wi-Fi during a medication pass is a patient safety issue, not an IT inconvenience. Migration must be invisible to the clinical user, which means thorough testing in a clinical simulation environment before production deployment.
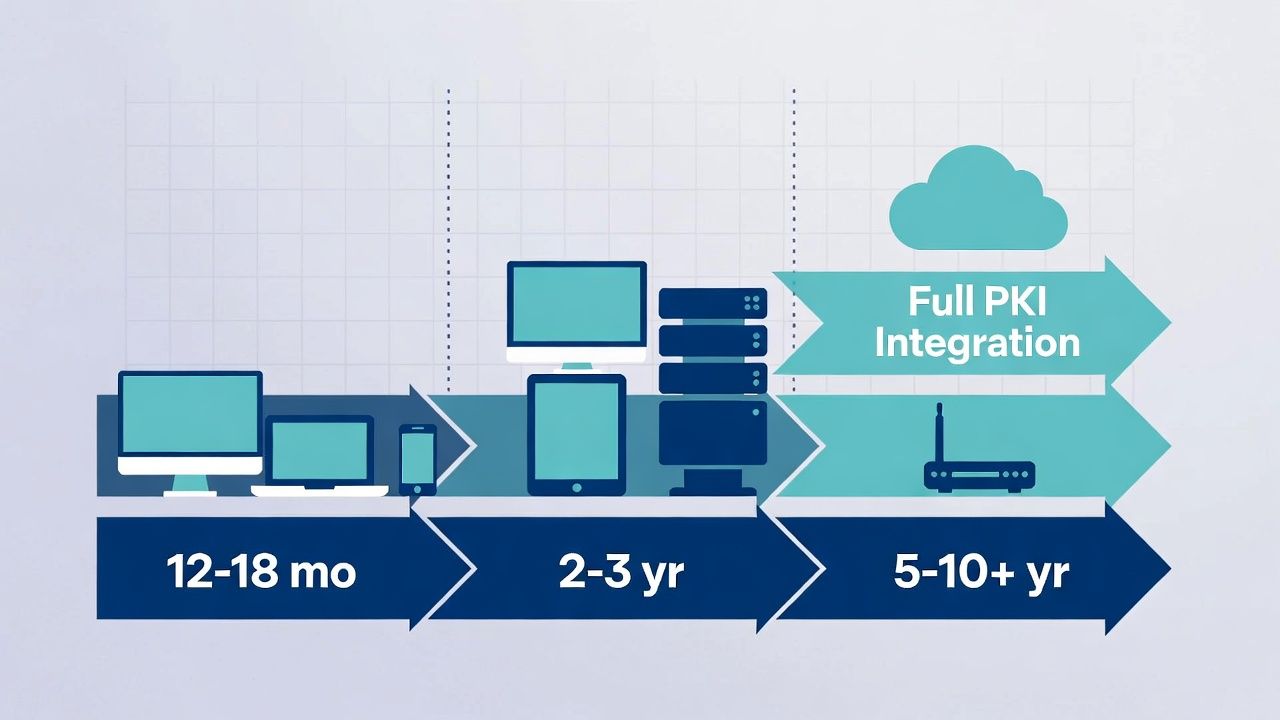
Migration Timeline Realities
Healthcare SCEP migration operates on three tracks simultaneously.
Fast track (12-18 months): Managed endpoints (clinical workstations, mobile devices) with MDM. Migrate to EST or ACME-based enrollment. This covers the largest volume of devices.
Medium track (2-3 years): Network infrastructure (switches, wireless controllers, VPN concentrators) where vendor firmware updates add modern enrollment support. Migrate as firmware updates are validated and deployed.
Slow track (5-10+ years): Medical devices with SCEP-only enrollment and no firmware update path. Contain, monitor, and accept the risk until device end-of-life replacement.
Related in this cluster: Certificate strategy hub · Private PKI · mTLS · M&A PKI · Multi-CA · Revocation · Certificate Transparency · CAA & DNS trust · Kubernetes TLS · Edge TLS · Code signing · Observability · SCEP / NDES sunset.